Organ transplantation: what it consists of, what the stages are and what the future holds
Organ transplantation is the surgical procedure whereby one or more diseased organs (whose functionality is no longer recoverable) are replaced with one or more organs taken from a donor (cadaveric or living)
An operation that has its roots, conceptually, in the earliest history of mankind (it was first spoken about by Chinese doctors), it is nevertheless a very recent therapeutic solution: the knowledge that made it possible (immunology, study of antigens…) was only acquired at the beginning of the 20th century.
From 1950 onwards, transplantation became an established choice in the treatment of those pathologies that lead to the irreparable destruction of the organ and, therefore, to the death of the patient.
But transplantation is not only the last prospect for those whose lives are in danger: this operation also makes it possible to improve the quality of life for those patients suffering from chronic disabling diseases (e.g. kidney transplantation for dialysed patients).
The future of transplantation is still to be sketched out, but is very clear in the minds of scientists and doctors engaged in research: implantation of artificial organs or organs taken from genetically modified animals (xenotransplantation), cloning and implantation of stem cells are just some of the directions in which the world’s scientific landscape is moving.
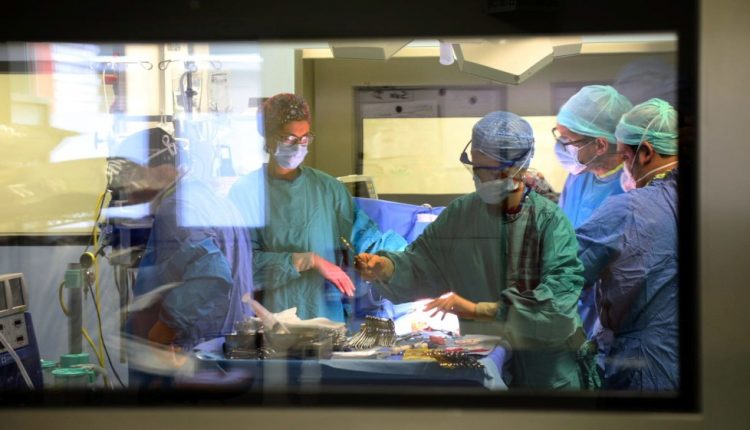
Organ transplantation surgery
The word ‘transplant’ often indicates, in a reductive way, the operation of replacing a diseased organ with a healthy one.
In reality, there is a whole organisation and preparation behind this operation that involves extreme precision and synchronisation of people and instruments.
The practice of the operation differs depending on the donor: if the organ removal is from a living person, in fact, it is possible to plan the operation; which is obviously not feasible if the organs come from a cadaveric donor, who died of accidental and unforeseeable causes.
Once the medical committee has obtained the family’s consent and declares the potential donor’s brain death to have occurred, the evaluation of his data begins: compatibility with potential recipients on the waiting lists, medical history, immune characteristics, blood group, etc.
Organ transplantation develops through several stages
PHASE 1
A person with injuries that could be a donor (for example, a very serious head injury) is admitted to intensive care.
A doctor speaks to the family about the possibility of donating his or her organs; if they are available, the coordination centre is immediately alerted, which is responsible for reporting the potential donor and identifying the potential recipient.
Meanwhile, the donor patient’s data are assessed: compatibility with potential recipients on the list, medical history, immune characteristics. The 6-hour observation period begins, which is mandatory before the certification of brain death.
PHASE 2
The explantation team is activated and must be available in a very short time.
The doctors usually reach the facility by helicopter. Meanwhile, at the hospital where the transplant will be performed, the recipient is called in to undergo various examinations and to assess his or her state of health.
Numerous checks are also carried out on the organs to be donated to prevent the transmission of infectious diseases or tumours from donor to recipient.
PHASE 3
At the end of the observation period, if all indications point to a diagnosis of irreversible brain death, explantation can begin (approximately 2 hours).
The recipient enters the operating theatre and is prepared for the operation. The administration of immunosuppressive drugs starts now to prevent the lymphocytes from recognising the organ as foreign and causing rejection.
PHASE 4
The organ finally arrives, immersed in a special solution to protect its cells and transported in a special container filled with ice to slow down its cellular activity.
One team of doctors prepares the recipient, the other takes care of cleaning the organ to be transplanted.
PHASE 5
The transplant can now begin: the blood vessels are connected, the bleeding is controlled.
STEP 6
The patient comes out of the operating theatre, but is still under anaesthesia, which will be prolonged for at least another 6 to 8 hours to allow the new organ to get used to the temperature difference between the container with the ice and the body and, of course, to the organ itself.
The patient remains connected to the machine to breathe.
STEP 7
The patient wakes up in the intensive care unit; if his general condition is good, he is taken off the artificial respirator.
After about 4 days, he starts walking again and eating.
After about 10 days, he will be able to leave the hospital and live with his new organ.
Initially, he will have to return to the hospital every day for immunological checks; after a year, he will be able to return once every two months.
Organ removal
Once brain death has been ascertained and the family’s consent obtained (in the case of a lack of explicit donor wishes), the potential donor is no longer assisted by the mechanical respirator and the organs can be harvested for transplantation in the same hospital that established suitability.
The previously alerted team enters the operating theatre for the removal operation.
Opposing the removal never means helping the patient to have better care; care, in fact, ends the moment brain death is established; opposing it would therefore only mean depriving someone else of a better life thanks to a new organ.
Today, another type of transplant is also gaining ground, that from living people.
Indeed, it is now possible to take a kidney, liver or lung lobe for transplantation in particularly at-risk people who would not survive on the waiting list.
These are usually children, both because of the shortage of paediatric transplant organs and because of the small size, which also means that the donor does not face too high a risk.
Once taken, organs require special procedures to preserve them for transplantation.
There is, for each organ, a maximum preservation time, beyond which the tissues, no longer receiving blood, and therefore oxygen, go into necrosis, i.e. their cells die, and are therefore unusable.
These times vary from organ to organ: heart (4-6 hours), lung (4-6 hours), liver (12-18 hours), kidney 48-72 hours, pancreas (12-24 hours).
Organ transplantation: rejection
Rejection is the reaction that the recipient organism has towards the transplanted organ or tissue.
In fact, the recipient’s immune system recognises the organ as foreign and attacks it as if it were a pathogen.
There are four types of rejection
- hyperacute rejection: this is the most rapid and occurs within a few minutes or hours after transplantation;
- accelerated rejection: often occurs in patients who have already received a previous transplant and occurs 3-4 days after the operation;
- acute rejection: occurs after a period of time ranging from 5 to 90 days; specific symptoms are oedema, fever, and loss of function of the transplanted organ;
- chronic rejection: develops about 3 months after transplantation and can cause tissue damage to the new organ to the point of loss of function.
Experiencing rejection of the transplanted organ does not necessarily mean inevitably losing it; on the contrary, rejection is successfully treated if action is taken within a reasonable time frame through the use of immunosuppressive drugs.
The immunosuppressants that the doctor prescribes after the transplant will help the transplanted organ not to risk rejection and to remain healthy.
Since the cells of the immune system are different, the drugs prescribed for immunosuppression will also be different.
Indications and contraindications for organ transplantation
The largest and most immediate indication for transplantation is irreversible failure of vital organs such as kidneys, liver, lungs, pancreas, but also corneas, bone marrow, intestines.
Indeed, in these cases, transplantation is the only effective treatment to ensure survival.
Therefore, any pathological condition that prevents the organ from functioning in such a way as to threaten the patient’s survival is to be considered an indication for transplantation.
Post-operative care
After transplantation, recipients are admitted for the first few days to a ward equipped for intensive care, where immunosuppressive therapy is started.
The immunosuppressed patient requires isolation in ‘sterile’ rooms, specially created to avoid contamination of any kind from the outside environment.
The ‘box’ in which the recipient is admitted after the transplant operation is completely isolated from the rest of the resuscitation unit used for conventional surgery.
The condition of strict isolation persists for as long as it takes for the patient to overcome the critical post-surgical phase (usually 5-6 days), or in cases where anti-rejection therapy is required.
Visits to transplant patients
In the immediate post-surgical period, visits to close relatives are permitted as long as they are appropriately dressed (according to the clean room entry procedures).
Each person is admitted to the filter zone one at a time and, of course, persons with suspicion and/or evidence of infectious diseases may not be admitted.
Future developments
The most serious issues in transplant medicine are, on the one hand, the rejection of the transplanted organ and, on the other, the insufficiency of donated organs compared to those needed.
In both directions, research is experimenting with various solutions to overcome these problems.
With regard to rejection, attempts are being made to create solutions that manage to trick the immune system, thus reducing the immunosuppressive therapy currently in use, or that protect the transplanted organ from attack by T lymphocytes, which are responsible for eliminating agents outside the body.
On the other front, that of organ shortage, artificial organs, tissue engineering or xenotransplantation are being experimented with that can replace human organs.
Gene therapy
Through gene therapy, it is possible to go to the source of the problem and eliminate genetic defects directly in the affected cells, tissues or organs.
The healthy gene is introduced directly into the affected spot, where it begins to produce those substances that the diseased body cannot produce on its own.
However, gene therapy is still far from being used. In order to be able to transport foreign DNA into the cell nucleus, special ‘vectors’ are needed – viruses that have lost their infectious characteristics, but are still able to attack cells and transmit their genetic heritage to them.
To avoid rejection, the organ to be transplanted would have to be treated in the laboratory, transferring genes into it that would make it capable of defending itself against the recipient’s immune system.
Now the genes are known, but they are not yet handled with the necessary precision. The next step will be to search for the perfect combination of genes that prevents the action of all the recipient’s immunological mechanisms.
Tissue engineering
The aim of this type of therapy is to find an alternative to human organs.
Already now, researchers are able to produce tissues such as blood vessels, heart valves, cartilage and skin in the laboratory.
It has been possible to overcome this new frontier thanks to the fact that cells tend to aggregate to form organs and tissues.
Stem cells
Stem cells are the undifferentiated cells found in human embryos one week after fertilisation.
They are also the ‘starting’ cells from which the tissues and organs of the child to be born will develop.
Their function is to regulate the turnover of blood cells (red blood cells, white blood cells and platelets) and those of the immune system (lymphocytes).
Today, computerised machines, separators, are used to collect these cells, allowing the selection of the necessary cells. The recipients of the cells are patients suffering from skin diseases, blood diseases or solid tumours.
In addition to the fact that stem cells are still largely unknown, there is also an ethical problem: harvesting embryonic stem cells implies the death of the embryo.
That is why the way to harvest stem cells from adults is being perfected.
Cloning
The cloning technique would make it possible to circumvent the problem of organ rejection altogether.
It would involve introducing the patient’s cell nucleus, with all its genetic heritage, into the stem cell of a human embryo or oocyte that previously had no nucleus of its own.
Cultivated in vitro in the laboratory, these modified cells would be genetically identical to those of the patient’s immune system, which would not recognise them as foreign.
This technique is not a viable option at present because both cloning, stem cell harvesting and the indiscriminate use of oocytes are prohibited by law.
Xenotransplantation
Xenotransplantation, i.e. the transplantation of animal cells, tissues and organs into humans, seems to be the future solution to the shortage of organs for transplantation.
Experiments in this field are numerous and face ethical, psychological and, last but not least, immune problems.
The few attempts that have been made, in fact (a pig liver and a baboon heart transplanted into two different human beings) have not yielded the desired results.
The rejection crisis, in fact, was particularly violent and impossible to control.
Yet this technique could really be the solution to the organ shortage.
In fact, what is most feared is the development of typically animal infections, transferred to humans via pathogens present in the organ to be transplanted, which could prove disastrous.
A possible alternative to this handicap could be genetic modifications on donor animals; in practice, the animals would be bred in a sterile environment and genetically modified to make their organs more compatible with the recipient’s organism.
For the time being, however, some milestones have been achieved; these are cell xenotransplants and not organ xenotransplants, such as pig embryo cells for the treatment of Parkinson’s disease, baboon marrow cells transplanted into terminally ill AIDS patients in an attempt to recover the patients’ immune system, or pancreas insulae still from pigs in the stimulation of insulin production as a therapy against diabetes.
Organ transplantation: artificial organs
Another solution to organ failure such as rejection is artificial organs.
The main problem is biological compatibility; these are, after all, mechanical organs that have to adapt to a biological organism.
Biocompatibility must cover all morphological, physical, chemical and functional characteristics that are able to provide for the organ’s functionality and, at the same time, its survival without the risk of rejection.
It is all these implications that make the production of artificial organs capable of completely and perfectly replacing ‘natural’ organs in their functions complex.
Read Also:
Emergency Live Even More…Live: Download The New Free App Of Your Newspaper For IOS And Android
Organ Transplantation: Diagnosis And Care Of Waiting Patients
What Is Cardiac Transplantation? An Overview
How Is A Face Transplant Performed? – VIDEO


